3D-Azide Microplates
for reaction with Alkyne groups or DBCO-modified molecules via Click chemistry
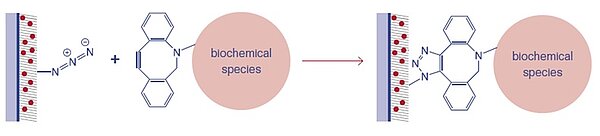
PolyAn’s 3D-Azide Microplates can be applied for bio-orthogonal binding of molecules that contain either Alkyne groups in a Copper-catalyzed Alkyne-Azide Cycloaddition, or Dibenzocyclooctyne (DBCO) groups in a Strain-promoted Alkyne-Azide Cycloaddition.
Products
PolyAn also equips glass slides, coverslips, polymer slides, and COP films with reactive 3D-surfaces. Please do not hesitate to contact us, if you would like to functionalize a different format or substrate with our 3D-Azide surface.
3D-Azide surface for DNA hybridization
Measurements to demonstrate and verify the efficient immobilization of DBCO-modified oligonucleotides to 3D-Azide microplates via copper-free click-chemistry.
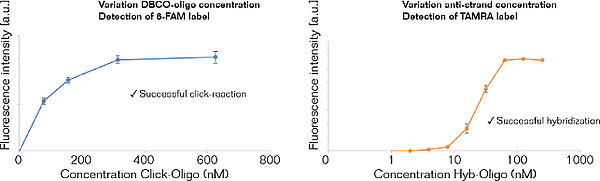
Left: Immobilization of a 5’-DBCO-oligonucleotide (20mer, 3'-labelled with 6-FAM) via strain promoted alkyne-azide cycloaddition (SPAAC), Right: Hybridization with the corresponding anti-strand (5’-labelled with TAMRA).
