Streptavidin and Neutravidin Surfaces
for non-covalent oriented coupling of Biotin modified biochemical species
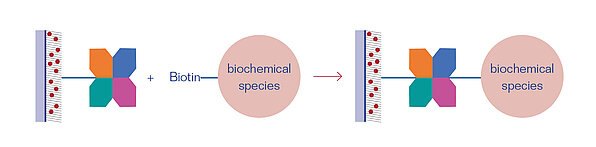
Streptavidin and Neutravidin are tetrameric proteins that can bind four Biotin molecules (vitamin B7) or any other Biotin-conjugated species with a very high specificity. The Streptavidin/Neutravidin-Biotin bond is one of the strongest, non-covalent bonds known in biochemistry, having a dissociation constant of KD = 10-15 mol/L. Thus, it is often applied in bioanalytical applications.
Streptavidin is a non-glycosylated protein purified from the bacterium Streptomyces avidinii. Neutravidin is a deglycosylated form of the native Avidin protein from egg white. Both Biotin-binding proteins can be distinguished by their isoelectric point, specificity, and non-specific binding:
| Avidin | Streptavidin | Neutravidin | |
|---|---|---|---|
| Molecular Weight | 67 kDa | 53 kDa | 60 kDa |
| Biotin Binding Sites | 4 | 4 | 4 |
| Isoelectric Point (pl) | 10 | 6.8-7.5 | 6.3 |
| Specificity | Low | High | Highest |
| Affinity for Biotin (KD) | 10-15 M | 10-15 M | 10-15 M |
| Nonspecific Binding | High | Low | Lowest |
Since PolyAn’s Streptavidin or Neutravidin matrices are covalently attached to the surface, the molecules are less susceptible to desorption in the presence of alkaline, acids, solutions of high ionic strength, or at high temperatures, compared to adsorptive immobilization.
PolyAn equips glass slides, coverslips, polymer slides and 96-well plates with reactive surfaces. Please do not hesitate to contact us, if you would like to functionalize a different format or substrate with our Streptavidin or Neutravidin surface.
Selected Publications
Streptavidin/Neutravidin surface for the immobilization of Peptides:
- Xuan, F. et al., `The Tudor-knot Domain of KAT5 Regulates Nucleosomal Substrate Acetylation´, J. Mol. Biol., 2024, 436, 168414. DOI: 10.1016/j.jmb.2023.168414.
- Hsu, C.-C. et al., `Gas41 links histone acetylation to H2A.Z deposition and maintenance of embryonic stem cell identity´, Cell Discovery, 2018, 4, 28. DOI: 10.1038/s41421-018-0027-0.
- Mi, W. et al., `The ZZ-type zinc finger of ZZZ3 modulates the ATAC complex-mediated histone acetylation and gene activation´, Nature Commun., 2018, 9, 3759. DOI: 10.1038/s41467-018-06247-5.
- Striffler, J.et al., `Replication of Polymer-Based Peptide Microarrays by Multi-Step Transfer´, Chem. Nano. Mat., 2016, 2, 897. DOI: 10.1002/cnma.201600194.
Streptavidin/Neutravidin surface for the immobilization of Glycans:
- Soto-Heras, S. et al., `Porcine sperm bind to an oviduct glycan coupled to glass surfaces as a model of sperm interaction with the oviduct´, Sci. Rep., 2025, 15, 4680. DOI: 10.1038/s41598-025-88986-2.
Streptavidin/Neutravidin surface for the immobilization of Proteins and Antibodies:
- Joshi, S. et al., `A versatile mitochondria isolation- and analysis-pipeline generates 3D nano-topographies and mechano-physical surface maps of single organelles´, bioRxiv, 2021, DOI: 10.1101/2021.10.31.466655.
