Surfaces for Click Chemistry
Click chemistry describes quick and irreversible one pot conjugation reactions that have a high reaction specificity, high yield of the desired product, and only minimal and inoffensive byproducts. Bio-orthogonal reactions are conjugation reactions that do not interfere with biological processes. Such reactions are especially useful in chemical biology, as they can be conducted under physiological conditions and address the need for highly specific and robust reactions in biological contexts.
PolyAn offers a variety of surfaces that are suitable for bio-orthogonal conjugation of biomolecules via click chemistry.
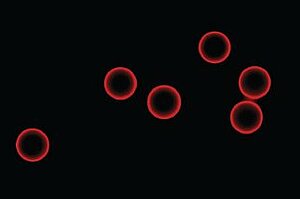
Transparent PolyAn microparticles with 3D-Alkyne surface coupled to fluorescence labeled azido peptide via ”click chemistry”.
[Fluorescence microscope image: Olympus IX71, 60x: visualisation of positive ligand binding]
3D-Azide
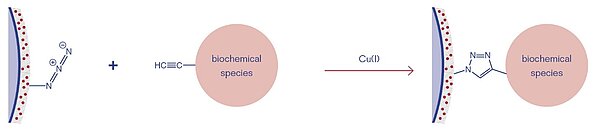
3D-Alkyne
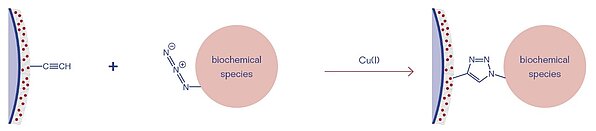
3D-DBCO

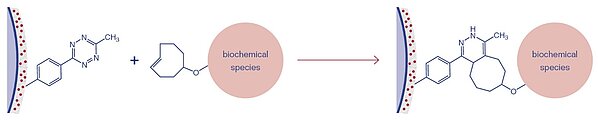
3D-MTZ
Key features
- Alternative to conventional Streptavidin-Biotin coupling
- Less unspecific interactions compared to Streptavidin
- No reactions of Alkynes, Azides, DBCO, or MTZ with regular biomolecule functionalities, e.g. Hydroxy, Amino, and Carboxy groups
PolyAn can functionalize all the beads from our portfolio with the new 3D-Azide, 3D-Alkyne, 3D-MTZ and 3D-DBCO surfaces, respectively:
- Transparent beads
- Fluorescence encoded beads
- Multiplex bead populations with up to 25 peaks for flow cytometry and fluorescence imaging systems
As part of our functionalization services PolyAn is now also offering the custom modification of beads with oligonucleotides or peptides as well as 3D-MTZ (Methyltetrazine) and 3D-DBCO (Dibenzylcyclooctyne) surfaces, respectively. Please do not hesitate to contact us (mail@poly-an.de), if you have any questions regarding our products. We are looking forward to your inquiry.
